| Starpharma |
| Registration Date | 10 Sep 2022 |
| Revision Date | 10 Sep 2022 |
| Share |
Medicine Pharmaceuticals
Nasal Spray
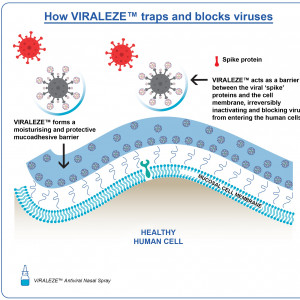
VIRALEZE™ has been developed by Starpharma, a world leader in the development of new medical products based on its proprietary dendrimer technology, as an antiviral nasal spray that is intended to provide a moisturising and protective barrier in the nose. VIRALEZE™ also contains SPL7013, which has been shown in laboratory studies to inactivate a broad spectrum of respiratory/cold viruses.
The nasal cavity has been shown to be a primary site where respiratory viruses, including cold viruses and SARS-CoV-2 (the virus that causes COVID-19), become established, before spreading in the body (Hou et al., 2021).
SPL7013 was originally developed by Starpharma for its antiviral activity and has been shown to block a broad-spectrum of viruses in nonclinical in vitro and in vivo studies (Bernstein et al, 2003; Jiang et al, 2005; Gong et al, 2005; Lackman-Smith et al, 2008; Tyssen et al, 2010; Telwatte et al, 2011; Romanowski et al, 2021).
Starpharma subsequently generated and published laboratory data showing that SPL7013 also inactivates >99.9% of coronavirus SARS-CoV-2 (Paull et al., 2021a).
Starpharma has also published data demonstrating the protective efficacy of VIRALEZE™ antiviral nasal spray against SARS-CoV-2 challenge in vivo in a humanised mouse model of coronavirus infection. The study results showed that VIRALEZE™ administered nasally reduced viral load by >99.9% (vs. saline control) in the lungs and trachea of animals challenged with SARS-CoV-2. The protective effects of VIRALEZE™ against SARS-CoV-2 in animals are consistent with the previously reported in vitro virucidal activity of SPL7013, which reduces infectious SARS-CoV-2, including the Delta variant, by >99.9% within 30 seconds of exposure. The results of the in vivo study (conducted at The Scripps Research Institute) have been published in the peer-reviewed journal, Viruses, in a special issue titled, Medical Interventions for Treatment and Prevention of SARS-CoV-2 Infections (Paull et al., 2021b).
SPL7013 has also been shown in laboratory studies conducted at the Scripps Research Institute to inactivate several SARS-CoV-2 “Variants of Concern”, including the Alpha, Beta, Gamma, Delta and Omicron variants, with testing planned for other variants as they emerge and become available.
In addition, Starpharma announced that SPL7013 was shown in laboratory studies to block human respiratory syncytial virus (HRSV), and to block infection by other pandemic coronaviruses (SARS-CoV, MERS-CoV) and influenza viruses. On 18 May 2022, Starpharma reported new virucidal data for SPL7013 against influenza virus A and B, in laboratory studies. SPL7013 achieved 95% and 99.7% reduction in viral infectivity against influenza virus A and B, respectively.
SPL7013 is a patented, precisely designed large negatively charged molecule that has been shown to not pass into the bloodstream following topical application to mucosal epithelial surfaces. In VIRALEZE™, SPL7013 is intended to form a barrier locally at site of application in the nasal cavity to physically trap and thereby inactivate respiratory viruses.
Starpharma was awarded development funding for the development and commercialisation of VIRALEZE™ by the Australian Government’s Medical Research Future Fund Biomedical Translation Bridge Program.
VIRALEZE™ is registered for sale in more than 30 countries, including in Europe, Asia, and the Middle East. VIRALEZE™ launched in the UK through LloydsPharmacy, as announced on 25 March 2021 and then in Europe via Starpharma's webstore, as announced on 6 May 2021. VIRALEZE™ has also launched in Vietnam as announced on 3 December 2021. Starpharma is also progressing regulatory activities for a number of markets, including Australia, with the intention of rolling it out across other markets. VIRALEZE™ is not approved for sale or supply in Australia.